About SARS-CoV-2 (COVID-19) testing
COVID-19 is an emerging infection and there is still much to learn about the mechanism about Covid-19 measurement. Nanjing Norman Biological Covid-19 solution, providing different Covid-19 testing kits to measure the antigen or antibody incurred by covid-19 is now available.
The information these tests provide is essential to monitor and control the infection, and the development of effective therapeutic treatment and vaccines.
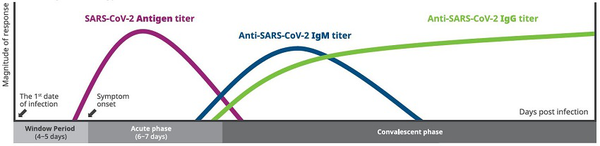
Time Kinetics of Antigen and Antibody Response in SARS-CoV-2
Time kinetics of antibody response in coronavirus disease 2019 (COVID-19). The illustration demonstrates the relative levels of ARS-CoV-2 Antigen and host immunoglobulins (IgM, IgG) at different stages of COVID-19.
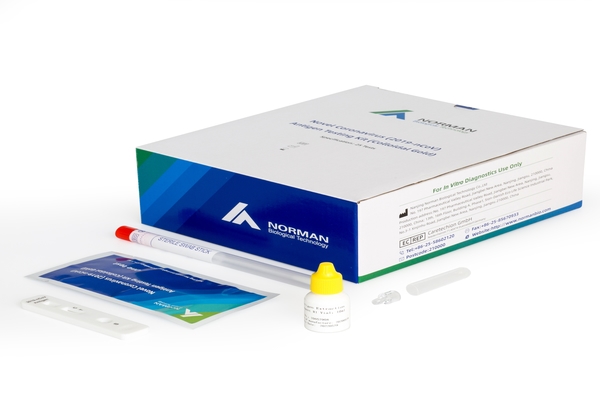
Why choose Norman Novel Coronavirus (2019-nCoV) Antigen Testing Kit (Colloidal Gold)
Virus detection in the laboratory is performed on nasopharyngeal and throat swabs using molecular PCR tests. PCR is the most sensitive method for detecting the virus early upon infection, sometimes before the onset of symptoms, and requires the use of specialized analyzers in a laboratory, often taking several hours to run the test.
From a patient’s point of view, rapid antigen testing works in much the same way as molecular testing. Your health care provider will swab the back of your nose or throat to collect a sample for testing. But instead of waiting days for your results, an antigen test can produce a result in an hour or less, says the FDA.
Norman biological COVID-19 Antigen kits are designed for the rapid detection of SARS-CoV-2 antigen in nasopharyngeal and throat swabs OR the saliva,final result takes 15mins .
These kits are for professional use and are intended as a screening test to aid in the early diagnosis of SARS-CoV-2 infection in patients with clinical symptoms. Positive results should be considered in conjunction with the clinical history and other data available to the clinician.
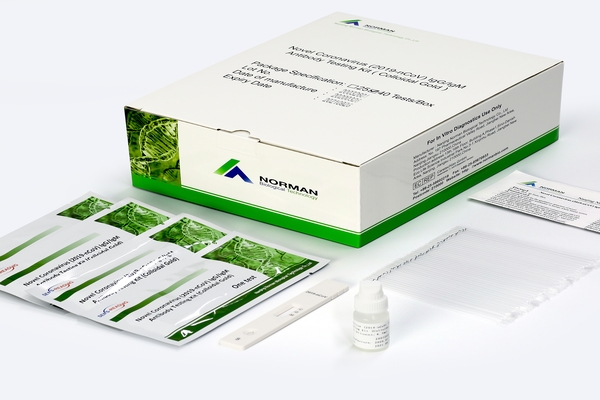
Why Novel Coronavirus (2019-nCoV) IgG/IgM Antibody Testing Kit (Colloidal Gold)
The detection of antibodies to SARS-Cov-2 in whole blood, serum or plasma samples is an indication of previous infection with the virus. As information about the antibody response to this virus increases, the clinical utility of laboratory and point of care tests will become clearer.
Serological testing is a useful tool in the surveillance of the infection and in determining the extent of the COVID-19 pandemic. It may also be helpful for the diagnosis of patients with a negative PCR result or for the identification of patients that have had asymptomatic infections.
Norman biological COVID-19 IgM/IgG Antibody rapid test is available for point of care antibody detection in whole blood from a finger-prick sample. These kits are not designed for home use and are available for professional use only in the global base. The results should be interpreted in conjunction with the clinical history of the individual, any previous exposure to the infection and the onset of any relevant symptoms. It has been shown that seroconversion rates are variable and it is recommended that samples for antibody detection are collected at least 2-3 weeks after onset of symptoms.
Get more information from www.normanbio.com.
Data sheet:
https://www.normanbio.com/js/htmledit/kindeditor/attached/20210127/20210127165753_83816.pdf
https://www.normanbio.com/js/htmledit/kindeditor/attached/20210116/20210116105841_49888.pdf
https://www.normanbio.com/js/htmledit/kindeditor/attached/20210302/20210302160245_14732.pdf
https://www.normanbio.com/js/htmledit/kindeditor/attached/20210302/20210302160353_87734.pdf
Prev: Why Medical Hand Towels Are Essential for Healthcare Settings
Next: Neutralizing antibodies – the immune system's Superhero